

2019年6月的SAT考试临近了,同学们准备好了吗?
北京新东方马聪老师今天就跟大家说说,SAT2化学考试中哪些知识点是不用看的。
2018年College Board更新了十年未曾更新的SAT2化学官方指南。其中最最重要的部分莫过于对考试范围的重新划定。有些以前认为考,但在新的SAT化学考试中不会出现的知识点,究竟是哪些呢?
1. 原电池与电解池
2. 吉布斯自由能
3. 杂化轨道理论
4. Lewis酸碱
5. 基元反应与非基元反应
以上是经常觉得需要复习但不会出现的知识点,有没有瞬间感觉舒爽了很多,下面再说说SAT2中需要格外注意的知识点。
大家都知道,SAT2是比较基础的考试,因此对于较深的理论讨论不多,反倒是对于一些常识内容做了要求,以下强调两点“常识”。
01 化合物的命名
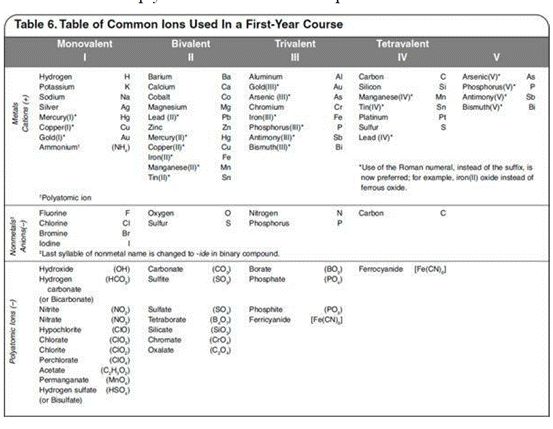
长话短说。关于命名,我们可以简单分为共价化合物的命名与离子化合物的命名。大家的命名规则都是先命化合价为正的,后命化合价为负的,区别是离子化合物中金属阳离子如果有多种价态,需用罗马数字标明价态,如氯化亚铜CuCl命名为Copper(I) chloride;共价化合物的命名需注意将化合价为负的原子的个数标明,作为化合价为负原子的前缀,如NO2 ---Nitrogen dioxide。表示个数的前缀分别为:mono(1),di(2),tri(3),tetra(4),pent(5)。此外还要注意酸碱的命名,下面给大家附一张各原子离子的命名表,不知道怎么命的时候可以掏出来看看。
02 实验
关于实验的部分,可考的方向真是很多呢,还好考得不多,只有8%(6-7道题)。给大家指出一些要点。
1)实验仪器:大家要做到考试中出现的实验仪器要认识,知道是干什么用的。常见仪器如下:

2)焰色反应:这种题如果出,也就一两道,大家需要知道常见金属燃烧的火焰颜色。常见如下:
Red: Lithium, Strontium
Orange: Calcium
Yellow: Sodium
Green: Barium
Violet: Potassium
3)沉淀:不同金属离子沉淀的颜色,以硫化物为例如下:
Lead (Pb): Brown-black (PbS)
Copper (Cu): Black (CuS)
Silver (Ag): Black (Ag2S)
Mercury (Hg): Black (HgS)
Nickel (Ni): Black (NiS)
Iron (Fe): Black (FeS)
Cadmium (Cd): Yellow (CdS)
Arsenic (As): Light yellow (As2S3)
Antimony (Sb): Orange (Sb2S3)
Zinc (Zn): White (ZnS)
Bismuth (Bi): Brown (Bi2S3)
4)实验原理:掌握过滤(Filtration)、蒸馏(Distillation)、色谱(Chromatography)、滴定(Titration)的原理。
5)注意事项:实验中操作层面的注意事项,如水别往硫酸里倒什么的。仰赖大家素来的化学基础和初高中阶段基本实验常识。
最后附上考试大纲,用以自查,祝大家考试顺利~
考纲
Structure of Matter 25%
Atomic Structure: including experimental evidence of atomic structure, quantum numbers and energy levels (orbitals), electron configurations, periodic trends
Molecular Structure: including Lewis structures, three-dimensional molecular shapes, polarity
Bonding: including ionic, covalent and metallic bonds; relationships of bonding to properties and structures; intermolecular forces such as hydrogen bonding, dipole-dipole forces, dispersion (London) forces
States of Matter16%
Gases: including the kinetic molecular theory, gas law relationships, molar volumes, density, stoichiometry
Liquids and Solids: including intermolecular forces in liquids and solids, types of solids, phase changes, and phase diagrams
Solutions: including molarity and percent by mass concentrations, solution preparation and stoichiometry, factors affecting solubility of solids, liquids and gases, qualitative aspects of colligative properties
Reaction Types14%
Acids and Bases: including Brønsted-Lowry theory, strong and weak acids and bases, pH, titrations, indicators
Oxidation-Reduction: including recognition of oxidation-reduction reactions, combustion, oxidation numbers, use of reduction potentials
Precipitation: including basic solubility rules
Stoichiometry14%
Mole Concept: including molar mass, Avogadro’s number, empirical and molecular formulas
Chemical Equations: including the balancing of equations, stoichiometric calculations, percent yield, and limiting reactants
Equilibrium and Reaction Rates 5%
Equilibrium Systems: including factors affecting position of equilibrium (Le Châtelier’s principle) in gaseous and aqueous
systems, equilibrium constants, and equilibrium expressions
Rates of Reactions: including factors affecting reaction rates, potential energy diagrams, activation energies
Thermochemistry6%
ncluding conservation of energy, calorimetry and specific heats, enthalpy (heat) changes associated with phase changes and chemical reactions, heating and cooling curves, entropy
Descriptive Chemistry12%
Including common elements, nomenclature of ions and compounds, periodic trends in chemical and physical properties of the elements, reactivity of elements and prediction of products of chemical reactions, examples of simple organic compounds and compounds of environmental concern
Laboratory 8%
Including knowledge of laboratory equipment, measurements, procedures, observations, safety, calculations, data analysis, interpretation of graphical data, drawing conclusions from observations and data
最新热文推荐:
(责任编辑:Hui)
 选择城市
选择城市